The renal pelvis may present a variety of abnormalities,
which either arise primarily from sinus structures or extend into it secondarily from the adjacent cortex or retroperitoneum.
Thickening of the renal pelvis may have an inflammatory/infectious or neoplastic etiology and may be mucosal or submucosal.
The identification of additional radiological features besides wall thickening – lithiasis,
pyelonephritis,
alterations in renal parenchyma,
segmental or diffuse thickening or the presence of air,
retroperitoneal adenopathies or the association with clinical and analytical findings – facilitate accurate diagnosis.
In this study,
we describe patients with radiological alterations of the renal pelvis on CT and correlate these abnormalities with the clinical and pathological evolution.
FALSE THICKNESS OF THE RENAL PELVIC WALL
1.- Vascular attachment to renal pelvic wall
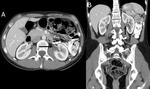
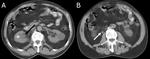
Vascular structures in the renal pelvis such as an artery or a vein may occasionally simulate a false increase in renal pelvic wall thickness. Fig. 3
PRIMARY ABNORMALITIES OF THE RENAL PELVIS
1.- INFLAMMATORY ETIOLOGY
Lithiasis
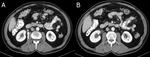
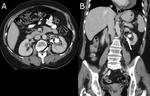
Impaction of kidney stones in the renal pelvic wall may cause inflammatory changes in the urothelial epithelium.
These CT images show a thick renal pelvic wall and radiodense stones. Fig. 4
Pyeloureteritis
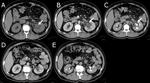
Pyeloureteritis is a rare entity that may simulate a urothelial neoplasm.
Diagnosis is based on clinical symptoms and laboratory findings.
In this case,
symptoms disappeared with antibiotic treatment.
Fig. 5 and Fig. 6
2.- INFECTIOUS ETIOLOGY
Bacterial pyelonephritis
In acute pyelonephritis,
the use of CT is usually reserved for patients whose clinical diagnosis is unclear,
patients who fail to respond to conventional medical treatment,
diabetics,
and other immunocompromised patients.
Bacterial renal infections,
except those caused by Gram-positive organisms,
occur via the ascending route and span a continuum of varying severity from uncomplicated acute pyelonephritis through progressively worsening stages of interstitial inflammation to frank abscess formation.
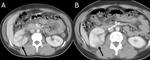
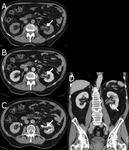
Wedge-shaped areas of hypoattenuating cortex and poor corticomedullary differentiation on a nephrographic CT scan is typical of acute pyelonephritis.
CT commonly demonstrates other signs of renal infection,
including soft-tissue stranding and thickening of the Gerota fascia,
obliteration of the renal sinus and calyceal effacement due to adjacent affected renal parenchyma,
thickening of the walls of the pelvis and calycesand mild dilatation of the renal pelvis Fig. 7 and ureter.
TBC pyelonephritis
The genitourinary tract accounts for 4–17% of extrapulmonary infections.
Urinary frequency,
pain,
hematuria,
and sterile pyuria may occur.
The upper renal tract is the most frequently affected part of the genitourinary tract,
and the renal parenchyma and upper collecting system are often simultaneously involved.
Renal parenchymal involvement can appear as focal nephritis,
nodular mass (tuberculoma),
or parenchymal scar formation.
Focal nephritis appears in the form of wedge-shaped areas of reduced enhancement that is identical in appearance to nephritis caused by other organisms.
Nodular mass is an uncommon finding that may be mistaken for a tumor.
Parenchymal scars and calcification can develop following healing by fibrosis.
The earliest change in the upper collecting system is papillary necrosis,
which may appear as small poorly defined areas of reduced enhancement at the tip of the medullary pyramids,
or as calyceal clefts and cavities.
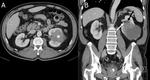
Late changes in the upper collecting system changes include wall thickening and fibrosis Fig. 8 .
Infundibular strictures can cause focal or uneven caliectasis.
Renal pelvis involvement may appear as a smooth,
angulated kink (Kerr’s kink) with generalized hydronephrosis or diffuse pelvic contraction.
Irregular ureteral thickening can show contrast enhancement and cause proximal hydro-ureteronephrosis.
End-stage tuberculosis can result in autonephrectomy,
in the form of a shrunken,
poorly functioning kidney with parenchymal thinning,
severe hydronephrosis,
and lobar calcification.
Lobar calcification occurs as a result of dystrophic calcification within caseous debris,
and it can form a cast of the kidney (putty kidney) when it is diffuse.
Advanced ureteral fibrosis causes a pipe-stem,
corkscrew,
or beaded appearance of the ureter owing to multiple strictures,
and mural calcification may occur.
Emphysematous pyelitis
Emphysematous pyelitis (EP) is a gas-forming urinary tract infection associated with gas localized exclusively in the renal collecting system.
This entity is cited in radiology textbooks and is associated with diabetes mellitus and obstruction of the collecting system.
However,
to our knowledge,
only two isolated cases have been described as case reports in the literature.
The diagnosis of EP is often delayed because clinical manifestation may be nonspecific and resemble that of uncomplicated acute pyelonephritis.
On radiographs,
the manifestation of this disorder is similar to findings on gas pyelograms,
with gas inside the pelvocalyceal system.
The gas may be present in the ureters and on rare occasions may be associated with emphysematous cystitis.
Although abdominal radiography usually allows easy detection of air,
the sensitivity reported with radiography is low (33%).
This is due to difficulty in differentiating renal gas from air in overlying loops of bowel.
In patients with diabetes and febrile urinary tract infection,
US is recommended for detecting urinary tract obstruction.
The typical US appearance of gas-producing renal infections comprises high-amplitude flat anterior margin echoes within the renal sinus or calyces,
which are associated with distal shadowing containing low-level echoes and reverberations.
In theory,
this dirty shadowing can be differentiated from the more distinct echo-free clean acoustic shadowing that occurs distally to renal calculi.
US provides less specific information about gas-producing renal infection because of the potential confusion with either air or renal calculi or calcifications within the kidney.
In practice,
confusion is possible and US images may be difficult to interpret.
CT is the most reliable diagnostic imaging technique currently available for this purpose.
Ureteral stones can be accurately evaluated and differentiated from intracalyceal,
intrapelvic,
or perinephric gas collections.
CT is a sensitive technique that is able to demonstrate and accurately locate air within the calyceal system,
thus allowing its elimination from/within the kidney parenchyma and/or pararenal spaces.
CT allows both accurate diagnosis and staging Fig. 9 .
Indeed,
EP should be differentiated from the reflux of air or gas from the bladder or from ileal ureterosigmoidostomy.
The spontaneous appearance of gas within the upper urinary tract has three main origins: iatrogenic manipulation (i.e,
ureteral instrumentation,
surgical or interventional radiologic procedure),
fistulous connection with a hollow viscus,
or bacteria caused by reflux from the bladder.
The overall mortality rate of EP is low.
If gas is localized in the collecting system and no obstruction is present,
antibiotic therapy alone appears to be sufficient.
CT can indicate the complete resolution of air and dilatation of cavities.
3.- NEOPLASTIC ETIOLOGY
Urothelial neoplasms
Transitional cell carcinoma (TCC) is commonly encountered in the urinary bladder and is usually diagnosed at cystoscopy.
Five per cent of urothelial tumors arise from the ureter or the renal pelvis or calyces,
accounting for approximately 10% of upper tract neoplasms.
Patients with TCC typically present with hematuria,
which may be frank or microscopic.
Up to one-third of patients present with flank pain or acute renal colic,
symptoms more typically associated with calculi.
Occasionally,
tumors may manifest with distant metastases or be discovered incidentally on radiologic examination.
Renal TCC most frequently arises in the extrarenal part of the pelvis,
followed by the the infundibulocalyceal region.
It is distributed equally between the left and right kidneys,
with 2%–4% of cases occurring bilaterally.
Twenty-five per cent of upper tract tumors occur in the ureter,
whereas 60%–75% of cases are found in the lower third,
with no side predominance.
Tumor spread occurs by mucosal extension or by local,
hematogenous,
or lymphatic invasion.
The most common sites for metastases are the liver,
bone,
and lungs.
The tumor stage at diagnosis influences the development of local recurrence and metastases and hence overall survival.
Multicentric TCC is common and is associated with poor survival.
Synchronous or metachronous tumor of the ipsilateral or contralateral collecting system is also common,
necessitating careful urologic and radiologic follow-up.
Upper tract TCC typically occurs in the sixth and seventh decades of life,
affecting males three times more often than females.
Besides increasing age and male gender,
the most important risk factor is smoking,
with smokers being two to three times more likely to develop TCC than nonsmokers.
Chemical carcinogens (aniline,
benzidine,
aromatic amine,
azo dyes),
cyclo-phosphamide therapy,
and heavy caffeine consumption are also associated with TCC,
and all predispose to synchronous and metachronous tumor development.
These substances are metabolized and excreted in the urine as carcinogenic substances which act locally on the urothelium.
Stasis of urine and structural abnormalities such as horseshoe kidney are also associated with increased prevalence.
These tumors are usually small at diagnosis,
grow slowly,
and follow a relatively benign course.
Pedunculated or diffusely infiltrating tumor is less common,
accounting for approximately 15% of upper tract TCCs,
but tends to behave more aggressively and be more advanced at diagnosis.
Infiltrating tumors are characterized by thickening and induration of the ureteral or renal pelvic wall.
If the renal pelvis is involved,
there is often invasion into the renal parenchyma Fig. 10 .
However,
this infiltrative growth pattern preserves the renal contour and differs from renal cell carcinoma,
which is typically expansile.
SECONDARY ABNORMALITIES OF THE RENAL PELVIS
1.- Renal neoplasms
Although renal collecting system invasion is not considered in the current TNM staging system of renal cell carcinoma,
this finding may be relevant in terms of treatment planning and prognosis.
Collecting system invasion exclusively in renal cell carcinoma with excretory phase CT images presenting a filling defect within the collecting system account for 5.4% of cases.
Fig. 11
2.- Lymphoma
Renal lymphoma appears in a wide variety of forms on CT.
In many cases,
diagnosis is not difficult because patients present with a known lymphoma at the time of imaging.
Lymphoma typically involves the kidney,
in one of several recognizable patterns including multiple renal masses,
solitary masses,
renal invasion due to contiguous retroperitoneal disease,
perirenal disease,
renal sinus involvement and diffuse renal infiltration.
No specific correlation has been found between the exact type of lymphomatous involvement and the pattern or prevalence of renal involvement.
Direct renal invasion from contiguous retroperitoneal disease is another common pattern of involvement in renal lymphoma,
and is seen in approximately 25%–30% of patients with documented disease.
These patients typically present with a large,
bulky retroperitoneal mass that envelops the renal vasculature and invades the renal hilum.
Lymphoma may affect the renal sinus,
although this is uncommon.
In most patients,
the renal arteries and veins remain patent despite tumor encasement.
However,
contiguous extension of retroperitoneal involvement in the renal collecting system can often cause obstruction,
and affected patients will commonly present with hydronephrosis Fig. 12 .
Transitional cell carcinoma is usually associated with a greater degree of obstruction of the collecting system Fig. 13 .
Displacement of the kidney can also be seen.
Following treatment of larger masses,
residual fibrosis is often seen and can be mistaken for recurrent or residual disease.